Introduction
Reserves of fresh water in the World reduce yearly and there is no hope to find new deposits of it. Of course, with the development of saving and purifying techniques of fresh water, the water crisis is delayed in time but not solved [1].
Another world-wide problem is lack of table salt. Many countries import the salt or produce from brines. On the other hand, it has proved futile to produce salt from sea water using current techniques as it was very laborious and ineffective. A hope of prevention of a shortage of fresh water and an increasing amount of consumption of table salt in countries which suffer from the acute shortage is exploitation of ocean water but problem is the lack of a suitable method of desalting that kind of water which requires minimal energy. This is crucial for world’s insufficient energy, especially in third world countries. Many laboratories work about desalination based on reverse osmosis [2, 3, 4, 5]. Desalination [6] performed by reverse osmosis involves the use of a synthetic semi-permeable membrane and a high pressure hydraulic membrane. Reverse osmosis is a physical process in which a proportion of water from a pressurized supply is forced through a semi-permeable membrane to become product water leaving behind almost all of the impurities in the remaining water. Ballester and Garrido [7] proposed the new desalination process idea involves the use of electrodialysis concepts regarding the use of charged-ion selective membranes to create differentiated cells of concentrated salt water and diluted or desalinized water. All of the processes need electric or fossil fuel energy, costly membranes, high pressure equipment and other components which make them useless for poor countries and additionally can pollute environment.
Liquid water is affected by magnetic fields [8]. Water is diamagnetic and may be levitated in very high magnetic fields (10 T) [9]. Weak magnetic fields (15 mT) have been shown to increase the evaporation rate of water solutions of NaCl, KCl, Na3PO4, and CaCl2 [10]. It is suggested that magnetic field treatment causes changes in the hydrating water structure around the ions. These effects are consistent with the magnetic fields weakening the van der Waals bonding between the water molecules and the water molecules being more tightly bound, due to the magnetic field reducing the thermal motion of the inherent charges by generating dampening forces [11].
The main aim of the experiments was researching influence of low magnetic fields between 0 and 0.8 T on water evaporation and crystallization of NaCl from saturated aqueous solutions of chemically pure NaCl.
Investigations of the influence of magnetic fields on crystallization compounds like CaCl2, (NH4)3PO4 and KH2PO4 were reported by Ma Wei et al. [12]. He concluded the magnetic field prompted the separation of tiny particles, increased the solubility of the phosphate and accelerated crystallization. The same process of the solution without magnetic field showed the crystallization speed was slower and the particles were larger.
Nyvlt and Krickova [13] showed that exposure of magnetic fields increased the nucleation kinetics during crystallization of MgSO4 from an aqueous solution. Mitrovic et al. [14] found that magnetic fields changed the growth rate distribution and decreased the growth rate of the majority of small Rochelle salt crystals from aqueous solutions. Higashitani et al. [15] investigated the effects of magnetic field on formation of CaCO3 crystals from the reaction between solutions of CaCl2 and Na2CO3. He noticed that the nucleation frequency of CaCO3 crystals suppressed but the growth of crystals was accelerated if the magnetic flux density was greater than about 0.3T and the exposure time is larger than 10 min; solutions exposed to a magnetic fields maintained the magnetic effect on the formation of CaCO3 for at least 120 h; formation of aragonite structure of CaCO3 crystals accelerated with the magnetic fields.
Theory of electrolyte crystallization from an aqueous solution of a sparingly soluble salt with NaCI structure in magnetic fields was described by Hans E. Lundager Madsen [16].
Reserves of fresh water in the World reduce yearly and there is no hope to find new deposits of it. Of course, with the development of saving and purifying techniques of fresh water, the water crisis is delayed in time but not solved [1].
Another world-wide problem is lack of table salt. Many countries import the salt or produce from brines. On the other hand, it has proved futile to produce salt from sea water using current techniques as it was very laborious and ineffective. A hope of prevention of a shortage of fresh water and an increasing amount of consumption of table salt in countries which suffer from the acute shortage is exploitation of ocean water but problem is the lack of a suitable method of desalting that kind of water which requires minimal energy. This is crucial for world’s insufficient energy, especially in third world countries. Many laboratories work about desalination based on reverse osmosis [2, 3, 4, 5]. Desalination [6] performed by reverse osmosis involves the use of a synthetic semi-permeable membrane and a high pressure hydraulic membrane. Reverse osmosis is a physical process in which a proportion of water from a pressurized supply is forced through a semi-permeable membrane to become product water leaving behind almost all of the impurities in the remaining water. Ballester and Garrido [7] proposed the new desalination process idea involves the use of electrodialysis concepts regarding the use of charged-ion selective membranes to create differentiated cells of concentrated salt water and diluted or desalinized water. All of the processes need electric or fossil fuel energy, costly membranes, high pressure equipment and other components which make them useless for poor countries and additionally can pollute environment.
Liquid water is affected by magnetic fields [8]. Water is diamagnetic and may be levitated in very high magnetic fields (10 T) [9]. Weak magnetic fields (15 mT) have been shown to increase the evaporation rate of water solutions of NaCl, KCl, Na3PO4, and CaCl2 [10]. It is suggested that magnetic field treatment causes changes in the hydrating water structure around the ions. These effects are consistent with the magnetic fields weakening the van der Waals bonding between the water molecules and the water molecules being more tightly bound, due to the magnetic field reducing the thermal motion of the inherent charges by generating dampening forces [11].
The main aim of the experiments was researching influence of low magnetic fields between 0 and 0.8 T on water evaporation and crystallization of NaCl from saturated aqueous solutions of chemically pure NaCl.
Investigations of the influence of magnetic fields on crystallization compounds like CaCl2, (NH4)3PO4 and KH2PO4 were reported by Ma Wei et al. [12]. He concluded the magnetic field prompted the separation of tiny particles, increased the solubility of the phosphate and accelerated crystallization. The same process of the solution without magnetic field showed the crystallization speed was slower and the particles were larger.
Nyvlt and Krickova [13] showed that exposure of magnetic fields increased the nucleation kinetics during crystallization of MgSO4 from an aqueous solution. Mitrovic et al. [14] found that magnetic fields changed the growth rate distribution and decreased the growth rate of the majority of small Rochelle salt crystals from aqueous solutions. Higashitani et al. [15] investigated the effects of magnetic field on formation of CaCO3 crystals from the reaction between solutions of CaCl2 and Na2CO3. He noticed that the nucleation frequency of CaCO3 crystals suppressed but the growth of crystals was accelerated if the magnetic flux density was greater than about 0.3T and the exposure time is larger than 10 min; solutions exposed to a magnetic fields maintained the magnetic effect on the formation of CaCO3 for at least 120 h; formation of aragonite structure of CaCO3 crystals accelerated with the magnetic fields.
Theory of electrolyte crystallization from an aqueous solution of a sparingly soluble salt with NaCI structure in magnetic fields was described by Hans E. Lundager Madsen [16].
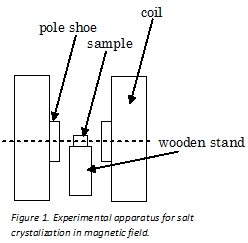
Experimental procedure
Saturated chemically pure NaCl solution in deionized water of volume 10 cm3 in glass vessels were putted between pole shoes of water cooled electromagnet on a wooden stand for thermal isolation (Figure 1).
A reference sample were placed in this same clean-room 4 meters from electromagnet where magnetic field was equal to background magnetic field. Temperature in the clean-room during all experiment was equal 20 deg C. Temperature between pole shoes was equal to temperature of environment and has not changed during time of working electromagnet. Vessels were covered by aluminium foil but not tightly and water vapour could move to environment. Samples crystallized in 0, 400 and 800 mT.
Obtained salt crystals were researched by X –ray diffraction (XRD) and scanning electron microscopy (SEM).
Saturated chemically pure NaCl solution in deionized water of volume 10 cm3 in glass vessels were putted between pole shoes of water cooled electromagnet on a wooden stand for thermal isolation (Figure 1).
A reference sample were placed in this same clean-room 4 meters from electromagnet where magnetic field was equal to background magnetic field. Temperature in the clean-room during all experiment was equal 20 deg C. Temperature between pole shoes was equal to temperature of environment and has not changed during time of working electromagnet. Vessels were covered by aluminium foil but not tightly and water vapour could move to environment. Samples crystallized in 0, 400 and 800 mT.
Obtained salt crystals were researched by X –ray diffraction (XRD) and scanning electron microscopy (SEM).

Results and discussion
Crystallization of reference sample (B=0mT) proceeded 11 day, sample in 400 mT – 6 days and sample in 800 mT – 7 days. Crystallized salt in 800mT is shown in figure 2.The first and the second part of the experiment confirmed Holysz et al. [17] conclusion that week magnetic field accelerates water evaporation and even increases crystallization ratio of unsaturated and saturated NaCl solutions.
The third part of the experiment showed surprising results.
The result showed that a dependence of salt crystallization and water evaporation on magnetic fields was not a monotone function. It suggested that salt crystallizes fastest in an optimal magnetic field around 400 mT.
Crystallization of reference sample (B=0mT) proceeded 11 day, sample in 400 mT – 6 days and sample in 800 mT – 7 days. Crystallized salt in 800mT is shown in figure 2.The first and the second part of the experiment confirmed Holysz et al. [17] conclusion that week magnetic field accelerates water evaporation and even increases crystallization ratio of unsaturated and saturated NaCl solutions.
The third part of the experiment showed surprising results.
The result showed that a dependence of salt crystallization and water evaporation on magnetic fields was not a monotone function. It suggested that salt crystallizes fastest in an optimal magnetic field around 400 mT.
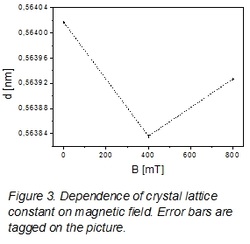
XRD analysis indicated for lattice constant modifications dependent on magnetic fields crystallization. The lattice constants were calculated by program Fullprof and they were: 0.56402nm for the reference sample (0mT), 0.56384nm – for 400mT and 0.56393nm – for 800mT with 0.00002nm of error (Figure 2). As it is shown in the figure 2 the smallest value of lattice constant is for 400mT of magnetic field and biggest is for 0mT. Of course, the difference between 0 mT and 400 mT was only 0.03% but error of calculation was less than 0.004%, what indicated on a contraction of lattice constant during crystallization in magnetic field. The broadening of spectral lines was observed as well. The calculated FWHM were 0.082, 0.103 and 0.088 for 0 T, 400 mT and 800 mT, respectively (Figure 4).

SEM pictures of the crystals (Figure 5) show the most regular structure for salt crystallized in 400 mT magnetic field and the most chaotic for salt in 0 mT.
Existence of the optimal, small magnetic field for evaporation water and crystallization NaCl can lead to great technical consequences. Usage magnetic fields in ocean water desalting system leads to larger desalting efficiency and allows save heat power for heating water to increase evaporation during desalting process or electric power in reverse osmosis. Magnetic fields until 1 T are generated easily by permanent magnets. Application of permanent magnets instead of electromagnets allows economizing electric power. A model way of solving this kind device is shown in a figure 5. Salt water is delivered to removable polyethylene containers by pipes. The containers are surrounded by permanent magnets created magnetic field about 0.4 T in the containers. Water evaporates from the containers and condenses on polyethylene roof and drips into other containers as fresh water which is then transported into pipes. After some cycles full of salt removable polyethylene containers are exchanged and table salt is acquired. Mirrors are used for intensification of sunbeams.
Conclusion
The aim of the research was finding the influence of magnetic field on NaCl crystallization form saturated solution. Experiments confirmed conjectures of acceleration water evaporation and crystallization of NaCl solutions by magnetic field. Additionally researches showed its modifying effect on crystal lattice. It was shown a certain value of magnetic field (around 400 mT) for the fastest crystallization and smallest constant lattice.
The results provide direction for a new approach to shortage of fresh water and table salt in the world. If the experiment will be confirmed for ocean water, the method of getting fresh water and table salt will be revolutionized. Usage of permanent magnets in desalting systems allows to economizing of heat and electrical energies necessary for ocean water desalting process.
The aim of the research was finding the influence of magnetic field on NaCl crystallization form saturated solution. Experiments confirmed conjectures of acceleration water evaporation and crystallization of NaCl solutions by magnetic field. Additionally researches showed its modifying effect on crystal lattice. It was shown a certain value of magnetic field (around 400 mT) for the fastest crystallization and smallest constant lattice.
The results provide direction for a new approach to shortage of fresh water and table salt in the world. If the experiment will be confirmed for ocean water, the method of getting fresh water and table salt will be revolutionized. Usage of permanent magnets in desalting systems allows to economizing of heat and electrical energies necessary for ocean water desalting process.
References
[1] Rogers Peter P., Scientific American, August (2008) 46-53 [2] M. Wilf, K. Klinko, Desalination 117 (1998) 323-331[2] M. Wilf, K. Klinko, “Effective new pretreatment for seawater reverse osmosis systems”, Desalination 117 (1998) 323-331
[3] M.Abraham, “Nanotech Water Desalination Membrane”, Nanotechnology Today, Nov 06, 2006
[4] Wasi Z.Khan “Desalination of raw water using a polyamide hollow fiber membrane”, Desalination 244 (2009) 59–65
[5] R. Verde, Equipment for desalination of water by reverse osmosis with energy recovery US patent application no 2001/0017278 (2001)
[6] Wasi Z.Khan, Desalination 244 (2009) 59–65
[7] P. Peñas Ballester, F. J. A. Garrido, International Conference on Renewable Energies and Power Quality (ICREPQ’09), Valencia (Spain), 15th to 17th April, 2009
[8] X.F. Pang and B. Deng, Science in China Series G-Physics Mechanics Astron, 51(2008) 1621-1632
[9] Y. Ikezoe, N. Hirota, J. Nakagawa and K. Kitazawa , Nature 393 (1998) 749-750
[10] L. Holysz, A. Szczes and E. Chibowski, , J. Colloid Interface Sci. 316 (2007) 996-1002
[11] H. Inaba, T. Saitou, K. Tozaki, and H. Hayashi, , J. Appl. Phys. 96 (2004) 6127-6132
[12] Ma Wei, Guo Liyan, Liu Xuehu, Yu Jie, ChJI, 2000 Vol.2 No.11 (2000) 52
[13] Nyvlt, J., Krickova J., Chem. Techn., 28 (1976) 548-550
[14] Mitrovic, M. M. et al., J. Crystal Growth, 87 (1988) 439-445
[15] Higashitani, K. et al., J. Colloid Interface Sci., 156 (1993) 90-95
[16] Lundager Madsen Hans E. Journal of Crystal Growth, 305, no. 1 (2007) 271-277
[17] L. Holysz, A. Szczesny and E. Chibowski, “Effects of static magnetic field on water and electrolyte solutions”, J. Colloid Interface Sci. 316 (2007) 996-1002.
[1] Rogers Peter P., Scientific American, August (2008) 46-53 [2] M. Wilf, K. Klinko, Desalination 117 (1998) 323-331[2] M. Wilf, K. Klinko, “Effective new pretreatment for seawater reverse osmosis systems”, Desalination 117 (1998) 323-331
[3] M.Abraham, “Nanotech Water Desalination Membrane”, Nanotechnology Today, Nov 06, 2006
[4] Wasi Z.Khan “Desalination of raw water using a polyamide hollow fiber membrane”, Desalination 244 (2009) 59–65
[5] R. Verde, Equipment for desalination of water by reverse osmosis with energy recovery US patent application no 2001/0017278 (2001)
[6] Wasi Z.Khan, Desalination 244 (2009) 59–65
[7] P. Peñas Ballester, F. J. A. Garrido, International Conference on Renewable Energies and Power Quality (ICREPQ’09), Valencia (Spain), 15th to 17th April, 2009
[8] X.F. Pang and B. Deng, Science in China Series G-Physics Mechanics Astron, 51(2008) 1621-1632
[9] Y. Ikezoe, N. Hirota, J. Nakagawa and K. Kitazawa , Nature 393 (1998) 749-750
[10] L. Holysz, A. Szczes and E. Chibowski, , J. Colloid Interface Sci. 316 (2007) 996-1002
[11] H. Inaba, T. Saitou, K. Tozaki, and H. Hayashi, , J. Appl. Phys. 96 (2004) 6127-6132
[12] Ma Wei, Guo Liyan, Liu Xuehu, Yu Jie, ChJI, 2000 Vol.2 No.11 (2000) 52
[13] Nyvlt, J., Krickova J., Chem. Techn., 28 (1976) 548-550
[14] Mitrovic, M. M. et al., J. Crystal Growth, 87 (1988) 439-445
[15] Higashitani, K. et al., J. Colloid Interface Sci., 156 (1993) 90-95
[16] Lundager Madsen Hans E. Journal of Crystal Growth, 305, no. 1 (2007) 271-277
[17] L. Holysz, A. Szczesny and E. Chibowski, “Effects of static magnetic field on water and electrolyte solutions”, J. Colloid Interface Sci. 316 (2007) 996-1002.


 RSS Feed
RSS Feed
